
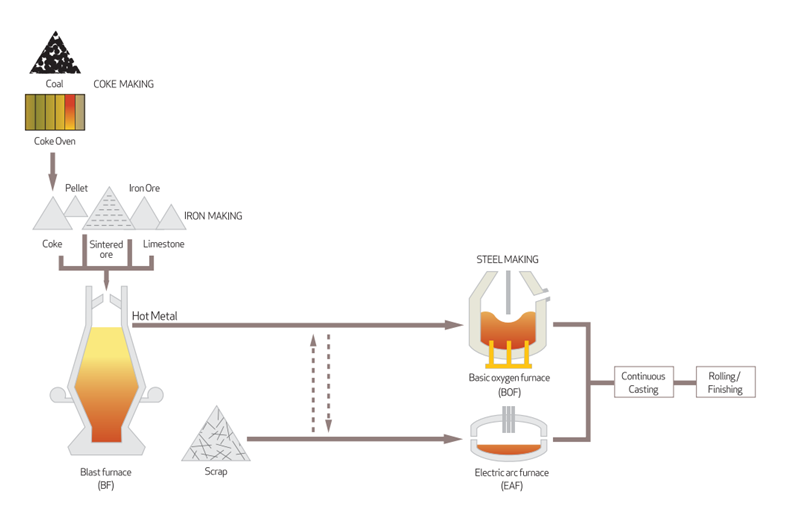
Coke is a solid, hard, porous and nearly 100% pure carbon material which is derived from heating coal in a coke oven to temperature as high as 20000C in order to drive out or liberate all the volatile constituents held inside the coal.
Volatile matter exists in coal primarily as organic material derived from the ancient fossilized vegetation. When liberated, the volatile material first appears as coal gas, light oil, and coal tar, which can be refined into coal chemicals as high-value by-products (e.g., toluene, benzene, phenol and numerous others).
Coke production is technology dependant. The traditional coke oven favored in commercial use captures the volatiles as by-products. The types and quality of coal that can be used are limited to a few. In the past 10 years, China has taken the lead to innovate and commercialize the no-byproduct, heat recovery coke-oven technology. One of the major advantages is opening up the possibilities of the type and quality of coal that can be blended for use as raw material for making coke.
Coke, produced in a traditional by-product recovery oven, has a typical relative density of ~0.78, is about 44% lighter than coal. Coke, produced from a no-by-product, heat-recovery coke oven, has a relative density of ~ 1.1 and about21% lighter than coal. Coke serves two main purposes: as a reducing agent to refine iron ore in a blast furnace to produce pig iron, and as a fuel to help to make it happen in the furnace.
Coke, limestone flux, and iron ore, and in that order, is filled or charged into from the top of the blast furnace, a tall, chimney-like structure. Charging the furnace with these raw materials is a very important step to control gas flow and chemical reactions inside the furnace. Hot air is blasted or blown into the bottom of the furnace to help the heating and combustion. Temperature in a blast furnace can reach as high as 1,3000 C to1,4000 C. Pig iron is the golden-color, hot, liquid metal that pours out of the bottom of a blast furnace. Pig iron is only an intermediate step to making other grades of iron (e.g., cast iron and wrought iron) and steel (e.g., carbon steel and alloy steel). Pig iron with its relatively high carbon content (~ 3.5 to 4.5%) is too brittle to be of much commercial use.
The entire process to produce pig iron, coke it is very important to possesses the right kind of physical and chemical properties. The words “thermally unstable” or “poor thermal stability” are often used to describe the characteristics of such poor quality coke. Coke also has to have little or no other impurities. To minimize any undesirable reactivity that may develop in the blast furnace, buyers look for coke with low sulfur, low ash, and low phosphorous. Depending on the specific applications, buyer may also try to avoid coke with other chemical constituents in the non-combustible portion of the ash.
In the commercial market place, coke is usually produced and sold as either foundry coke or metallurgical coke. In the trade, foundry coke refers to larger sizes of at least 80 mm and greater. Metallurgical coke is smaller in size; typically 45 mm to 60mm.
Foundry coke being larger is slower in reactivity as contrasted with the faster reactive metallurgical coke. Their usage depends on the end user’s applications. To produce coke with the above physical and chemical properties, the process begins with selecting the right kind of coal. In the trade, coal used to make coke is coking coal. In short; coke is one of important raw materials that is needed to produce pig iron in a furnace. Pig iron is used to make steel. Coke is used to extract iron from iron ore in a high-temperature furnace or oven. To make that happen, coke must be sufficiently porous and physically strong to withstand the violent chemical reactions in the furnace or oven, while coke itself is being consumed as fuel. And coke must be virtually free of impurities. The process all begins with selecting the right kind of coal as a coking coal in the first place.
正在加载,请稍候